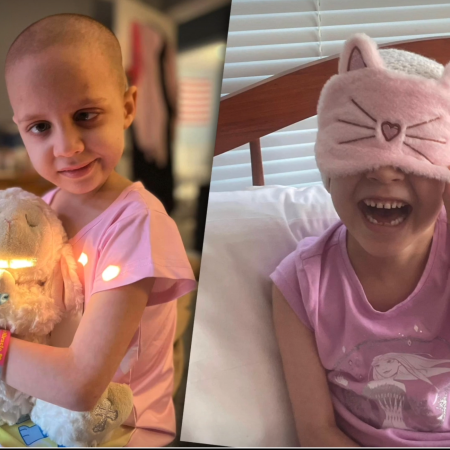
Diffuse Intrinsic Pontine Glioma (DIPG) is one of the most devastating childhood brain cancers, offering only temporary relief from current treatments. SonALAsense is developing a non-invasive sonodynamic therapy that targets tumor cells while sparing healthy brain tissue, giving children a safer and more effective treatment option. The next critical step is a pivotal clinical trial, and $7 million is needed to make this potentially life-saving therapy available to the children who urgently need it. Without this support, the therapy risks stalling in the “Valley of Death,” leaving families without hope. Your contribution can help advance this breakthrough and bring real options to children facing DIPG.
SHARE:
Diffuse Intrinsic Pontine Glioma (DIPG) is one of the most devastating childhood brain cancers. About 300 children in the U.S. are diagnosed each year, usually between ages 5 and 9. The tumors grow in a part of the brain that controls essential functions like breathing and movement, which makes surgery impossible. Current treatments, mainly radiation, can only offer short-term relief and often come with serious side effects that affect a child’s quality of life.
Families urgently need better options. SonALAsense is developing a new, non-invasive approach called sonodynamic therapy (SDT) that aims to target tumor cells while sparing healthy tissue. The treatment combines a drug derived from a natural nutrient with a special ultrasound device. The drug collects in cancer cells, and when activated by ultrasound, it helps destroy the tumor without harming surrounding brain tissue.
This targeted approach has the potential to provide children with DIPG a safer and more effective treatment than anything available today, giving families hope where it has been so desperately needed.
The next critical step is a pivotal clinical trial—the final stage needed to seek FDA approval for our sonodynamic therapy for DIPG. We are raising $7 million to make this trial a reality and bring this potentially life-saving, non-invasive treatment closer to the children who need it most.
We are in a strong position to reach this critical milestone. A lead investor has committed up to $7 million to support the pivotal trial, and the clinical-grade drug needed for a Phase III study has already been manufactured. We also have a strategic partnership with an ultrasound manufacturer to supply the devices for treatment.
Our experienced team has successfully guided multiple drug-device therapies through FDA approval, and we’ve built a strong foundation of support—including a $46.6 million ARPA-H award, a major federal investment in our technology. However, those federal dollars are limited to blood cancer research and cannot fund our DIPG trial.
Contributions through Music Beats Cancer fill this critical funding gap, helping unlock and complement these larger investments so this potentially life-saving therapy can move closer to FDA approval.
If this therapy were to disappear in the “Valley of Death,” it would mean something heartbreaking: another promising chance for children with DIPG—one of the most fatal childhood cancers—would be lost not because it failed, but because funding ran out. Over the past few years, early-stage support for rare pediatric diseases has dried up, and families have felt the impact firsthand. Therapies with real potential have stalled, and children continue to receive care that offers some comfort but no real hope of survival.
For families facing DIPG, time is often measured in months. They cannot wait for the funding landscape to rebound. Donor support is often the only force that keeps breakthroughs alive long enough to reach children and their families. Without it, we risk losing yet another opportunity to change the future for kids who desperately need more than the status quo.
SonALAsense: Developing a Safe and Effective Noninvasive Treatment for Cancer
ARPA-H funds SonALAsense to develop sonodynamic therapy to revolutionize treatment of blood cancers
Cancer Commons and SonALAsense: Putting Patients First
A Few Minutes with DIPG Nurse Navigator Leslie Jared
Dr. Stuart Marcus Creates SonALAsense To Treat Deadly Cancers
Father fights for new cancer treatment on NHS after death of actor son
SonALAsense: Developing a Safe and Effective Noninvasive Treatment for Cancer
Sonodynamic Therapy: An Emerging Treatment for Deadly Brain Tumors
Beating brain cancers with.. sound waves?
Historic First-in-Human Sonodynamic Therapy Clinical Trial for Glioblastoma
DIPG Patients using New Sonalasense Sono Dynamic Therapy with Insightec symptoms improve
Silent Warriors: United Voices Breaking the Silence on DIPG – Mark De Souza’s Story
Creating a cancer therapy without side effects
Co-Founder & Chief Executive Officer
Mark de Souza, PhD is a serial entrepreneur in the rare disease, oncology, and dermatology therapeutic areas. A biochemist by training, he spent 10 years in business development at Dyax Corp where he led or managed >50 licensing deals and was member of the Operations Committee that led the discovery and development of two drugs for hereditary angioedema. KALBITOR was FDA-approved in 2009. Shire acquired Dyax in 2015 for its second drug, TAKHZYRO (lanadelumab), which was FDA-approved in 2018. As an entrepreneur, Dr. de Souza founded and led Lotus Tissue Repair, which raised $26MM from Third Rock Ventures in 2011 to develop intravenous recombinant collagen VII as protein replacement therapy for dystrophic epidermolysis bullosa. Shire acquired Lotus in 2013. Dr. de Souza helped buy back the program from Shire and run clinical trials at a BridgeBio portfolio company called Phoenix Tissue Repair. Dr. de Souza became CEO of another BridgeBio company called PellePharm in 2015, and led the company through two Phase 2 trials with topical patidegib in Gorlin Syndrome, and obtained FDA breakthrough designation. The program is currently in Phase 3. Dr. de Souza is a co-founder of Molecular Skin Therapeutics, a BridgeBio portfolio company, developing a kallikrein 5/7 inhibitor for Netherton Syndrome. Dr. de Souza is a CoFounder & former Executive Chairman of NFlection Therapeutics, funded by FPrime Capital and venBio. NFlection has completed Phase 2 clinical trials with a topical MEK inhibitor for cutaneous tumors in neurofibromatosis. Dr. de Souza is a cofounder and CEO of SonALAsense.
Scientific Officer & Chief Medical Officer
Stuart Marcus, MD, PhD is a biochemist, medical oncologist, and expert in the clinical commercial development of drug-device combination therapies in the oncology and dermatology space. He entered the pharmaceutical industry in 1986 through Lederle Laboratories Medical Research Division, and became Group Director, Oncology where he developed Photofrin® (porfimer sodium) and laser light, the first drug+light combination photodynamic therapy (PDT) system. Dr. Marcus designed and carried out Phase II and III clinical studies on esophageal cancer, lung cancer, and bladder cancer, which led to the approval of this PDT system in multiple countries. He also led early clinical trials with PDT using Visudyne® (verteporfin) and laser light. Subsequently, Dr. Marcus became founding Chief Medical Officer of DUSA Pharmaceuticals, Inc., a public company, in 1993, charged with developing aminolevulinic acid (ALA) for the first topical PDT indications. He led the development of Levulan® (ALA) Kerastick® topical solution applicator and the BLU-U® blue light photodynamic therapy illuminator for the topical ALA PDT treatment of pre-cancerous skin lesions (actinic keratoses) through FDA approval in 2003. DUSA was purchased by Sun Pharma Industries in December 2012. Dr. Marcus is the scientific founder, Chief Scientific Officer & Chief Medical Officer of SonALAsense and PI of SonALAsense’s $46.6MM ARPA-H award to develop sonodynamic therapy in blood cancers.